 Hydrogen embrittlement of grain boundaries in nickel: an atomistic study (镍晶界的氢脆化:原子水平上的研究)
Hydrogen embrittlement of grain boundaries in nickel: an atomistic study (镍晶界的氢脆化:原子水平上的研究)
Shan Huang, Dengke Chen, Jun Song, David L. McDowell & Ting Zhu
npj Computational Materials 3:28 (2017)
doi:10.1038/s41524-017-0031-1
Published online:14 July 2017
Abstract| Full Text | PDF OPEN
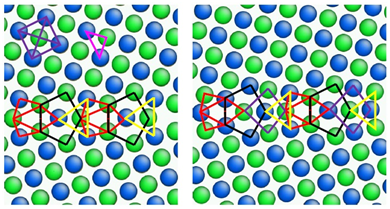
摘要:氢对金属的化学力学性能的削弱作用已有大量观察,但其原子尺度上的作用机理尚不清楚。本研究从原子水平上探讨了氢对镍晶界的脆化作用。通过将晶界划分为由原子组成的多面体堆积单元,可以确定出给定任意晶界的所有可能的氢间隙位点。计算得到各个间隙位点氢的偏聚能并代入Rice-Wang界面脆化热力学理论模型中。通过计算氢偏聚晶界的分离功的变化可以定量评估氢脆化效果。我们研究了快速和慢速分离极限,他们分别对应固定氢浓度和氢化学势条件下的晶界断裂。据此我们还进一步分析了局部电子密度对于氢吸附能的影响,从而揭示了晶界氢脆化的物理极限。
Abstract: The chemomechanical degradation of metals by hydrogen is widely observed, but not clearly understood at the atomic scale. Here we report an atomistic study of hydrogen embrittlement of grain boundaries in nickel. All the possible interstitial hydrogen sites at a given grain boundary are identified by a powerful geometrical approach of division of grain boundary via polyhedral packing units of atoms.Hydrogen segregation energies are calculated at these interstitial sites to feed into the Rice–Wang thermodynamic theory of interfacial embrittlement. The hydrogen embrittlement effects are quantitatively evaluated in terms of the reduction of work of separation for hydrogen-segregated grain boundaries. We study both the fast and slow separation limits corresponding to grain boundary fracture at fixed hydrogen concentration and fixed hydrogen chemical potential, respectively.We further analyze the influences of local electron densities on hydrogen adsorption energies, thereby gaining insights into the physical limits of hydrogen embrittlement of grain boundaries.
Editorial Summary
Metals: hydrogen probe divides nickel into grains(金属:氢使镍裂解为晶粒)
确定金属晶界处所有氢吸附位点,使金属材料失效的定量预测成为可能。由佐治亚理工学院的Ting Zhu领导的国际研究团队发展了一种方法,将氢原子嵌入金属镍晶格中并按照几何结构特征将金属镍划分成由原子组成多面体堆积单元,由此得到了所有可能的氢的吸附位点。通过对被氢吸附的晶界原子水平模拟,发现氢通过电子效应与镍结合并在较高温度下恶化、破坏晶界。晶界强度降低不仅取决于氢嵌入镍中的位置,也取决于晶界处的氢浓度。建立用于量化氢吸附造成金属失效的计算模拟框架将有助于减轻含氢压力容器和管道的失效。
Identifying all hydrogen absorption sites at metallic grain boundaries allows for a quantitative prediction of metal failure. An international team of researchers led by Ting Zhu at Georgia Institute of Technology developed a method to geometrically divide nickel metal into polyhedral packing units of atoms by embedding hydrogen atoms into the nickel atomic lattice. By using atomistic simulations to examine the boundaries through these atomic packing units, they showed that hydrogen binds to nickel via an electronic effect and that grain boundary failure due to hydrogen worsens at higher temperatures. Reducing grain boundary strength therefore depended on the location of hydrogen embedded in nickel as well as hydrogen concentration at the grain boundaries. Establishing a framework to quantify metal failure due to hydrogen absorption may help up mitigate failure in hydrogen-containing pressurized vessels and pipelines.


 沪公网安备 31010502006565号
沪公网安备 31010502006565号