 Identifying quasi-2D and 1D electrides in yttrium and scandium chlorides via geometrical identification(通过几何识别来筛选氯化钇和氯化钪中的准1D、2D电子化合物)
Identifying quasi-2D and 1D electrides in yttrium and scandium chlorides via geometrical identification(通过几何识别来筛选氯化钇和氯化钪中的准1D、2D电子化合物)
Biao Wan, Yangfan Lu, Zewen Xiao, Yoshinori Muraba, Junghwan Kim, Dajian Huang, Lailei Wu, Huiyang Gou, Jingwu Zhang, Faming Gao, Ho-kwang Mao & Hideo Hosono
npj Computational Materials 4:77 (2018)
doi:s41524-018-0136-1
Published online:17 December 2018
Abstract| Full Text | PDF OPEN
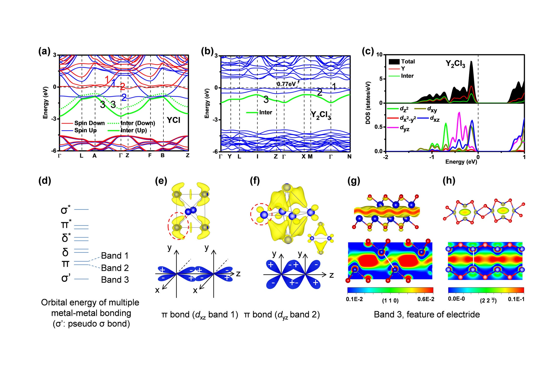
摘要:认识和理解富电子的电子化合物有望为各种电子和催化应用提供一个很有前途的机会。利用几何识别策略,本研究识别了一类新的电子材料:钇/钪氯化物Y(Sc)xCly (y:x<2)。阴离子电子存在于金属八面体骨架拓扑结构中。这些电子化合物的不同电子维度由准二维电子化合物和准一维电子化合物精确量化。其中,准二维电子化合物为 [YCl]+·e-和[ScCl]+·e-,准一维电子化合物为含有二价金属元素(Sc2+: 3d1和Y2+: 4d1)的[Y2Cl3]+·e-、[Sc7Cl10]+·e-和[Sc5Cl8]2+·2e-。具有局域化特征的阴离子电子被限制在内层空间,而不是在A2B-型二维电子化合物(如Ca2N)中观察到的层间空间中。此外,当氢原子被引入到框架结构中形成YClH和Y2Cl3H时,形成的相转变为传统的离子化合物,但由于费米能级能量增加,其功函数却出现了惊人的降低,这与迄今为止报道的常规电子化合物相反。实验证实Y2Cl3是带隙为1.14 eV的半导体。这些结果可能有助于促进新的电子化合物材料的理性设计和理性探索,为后续技术应用奠定基础。
Abstract:Developing and understanding electron-rich electrides offers a promising opportunity for a variety of electronic and catalytic applications. Using a geometrical identification strategy, here we identify a new class of electride material, yttrium/scandium chlorides Y(Sc)xCly (y:x<2). Anionic electrons are found in the metal octahedral framework topology. The diverse electronic dimensionality of these electrides is quantified explicitly by quasi-two-dimensional (2D) electrides for [YCl]+·e- and [ScCl]+·e- and one-dimensional (1D) electrides for [Y2Cl3]+·e-, [Sc7Cl10]+·e-, and [Sc5Cl8]2+·2e- with divalent metal elements (Sc2+: 3d1 and Y2+: 4d1). The localized anionic electrons were confined within the inner-layer spaces, rather than inter-layer spaces that are observed in A2B-type 2D electrides, e.g. Ca2N. Moreover, when hydrogen atoms are introduced into the host structures to form YClH and Y2Cl3H, the generated phases transform to conventional ionic compounds but exhibited a surprising reduction of work function, arising from the increased Fermi level energy, contrary to the conventional electrides reported so far. Y2Cl3 was experimentally confirmed to be a semiconductor with a band gap of 1.14 eV. These results may help to promote the rational design and discovery of new electride materials for further technological applications.
Editorial Summary
Electride materials: geometrical identification (电子化合物材料:几何识别)
从头算方法确定了一类稀土元素的电子化合物——钇/钪氯化物。来自东京理工大学高压科学与技术高级研究中心的Huiyang Gou和东京理工大学的Hideo Hosono领导的团队开发了一种基于几何识别和高通量从头算的材料筛选策略,确定了各种化学计量比的钇、钪氯化物及组合结构中新的准一维和准二维电子化合物。他们发现这些材料具有独特的特征,比如R-Cl的密堆积结构(R为Y或Sc)和八面体框架拓扑结构。仅Y2Cl3为一个半导体,实验测得的带隙为1.14eV,其它结构均具有铁磁性。当氢原子被引入该类结构中时,在插层化合物中观察到了功函数的显著降低。
Ab initio calculations identify a class of rare earth yttrium and scandium chloride electrides. A team led by Huiyang Gou at Center for High Pressure Science and Technology Advanced Research and Hideo Hosono at Tokyo Institute of Technology developed a materials screening strategy based on geometrical identification and high-throughput ab initio calculations to identify yttrium and scandium chlorides with various stoichiometries and compositions as a new class of quasi-2D and quasi-1D electrides. These materials were found to have distinctive features such as an R-Cl close-packed structure (R being Y or Sc), and an octahedral framework topology. The analyzed electrides were ferromagnetic, with the exception of Y2Cl3, which was found to be a semiconductor with an experimentally measured bandgap of 1.14eV. When hydrogen atoms were introduced in the structures, a reduction of work function was observed in the intercalated compounds.


 沪公网安备 31010502006565号
沪公网安备 31010502006565号